Lameness is a prevalent condition prompting pet owners to seek veterinary care, often leading to radiographic examination of the affected limb. Despite advancements in diagnostic imaging, radiography remains the cornerstone in evaluating lameness in small animal practice. Early radiographic evaluation is crucial in juvenile canine patients, especially larger breeds, where prompt identification of musculoskeletal pathology is essential because of the risk of serious growth-related complications. Delayed diagnosis and inappropriate treatment in certain conditions can result in abnormal bone growth, joint incongruity and irreversible osteoarthrosis (Fox, 2021). This emphasises the continued importance of radiography in the management of lameness in young patients.
This article discusses radiological aspects of key juvenile orthopaedic conditions for which timely diagnosis and intervention are beneficial for the patient. The discussion will focus on hip and elbow dysplasia, osteochondrosis, humeral intracondylar fissures, retained endochondral cartilage cores, hypertrophic osteodystrophy and physeal trauma. Emphasis will be placed on the importance of understanding patient signalment, disease prevalence in certain breeds, recognising normal anatomical variations and avoiding common pitfalls in radiographic interpretation.
Familiarity with normal radiographic findings and artifacts
Radiological assessment of juvenile patients poses challenges because of the presence of radiolucent physes, and as such, subtle physeal pathology may not be appreciable on the initial radiological evaluation. Several reference radiological texts are available (Coulson and Lewis, 2008; Thrall and Robertson, 2016) and comparative radiographs of the opposite limb, if this is normal, can also aid interpretation (Stander and Cassel, 2016). Obtaining orthogonal radiographs (two views at 90° to each other; Stander and Cassel, 2016), is essential for accurate interpretation and should be considered as the standard of care, after clinically identifying the site of lameness.
Physeal and apophyseal closure times and general anatomy
It is useful to be familiar with, or to consult published references (Dennis et al, 2010a; Stander and Cassel, 2016) with regards to the expected age ranges of appearance and closure times of physes and apophyses, and to know where these may occur. For example, knowing that the supraglenoid tubercule of the scapula has a separate centre of ossification that closes at approximately 4–7 months of age (Dennis et al, 2010a) will prevent an erroneous diagnosis of an avulsion fracture, in conjunction with a clinical examination. Being aware that a separate centre of ossification of the anconeus process only occurs in large breed dogs, and is radiologically first visible at 6–8 weeks, fusing to the ulna by 5 months of age (Dennis et al, 2010a; Vezzoni and Benjamino, 2021) is a requirement for accurate assessment of the juvenile elbow, with regards to elbow dysplasia.
Metaphyseal cutback or cutback zones
In young dogs, bone remodels from the physes towards the diaphyseal cortex, and the metaphyseal cortical margins may be irregular as a result (Nap and Hazewinkel, 1994; Nelson, 2023) (Figure 1). This is known as the ‘cutback zone’ and is a normal finding, especially in large and giant breeds, with the distal radius and ulna often demonstrating the most obvious change. Care should be taken not to confuse these regions for pathological periosteal reactions or osteolysis.
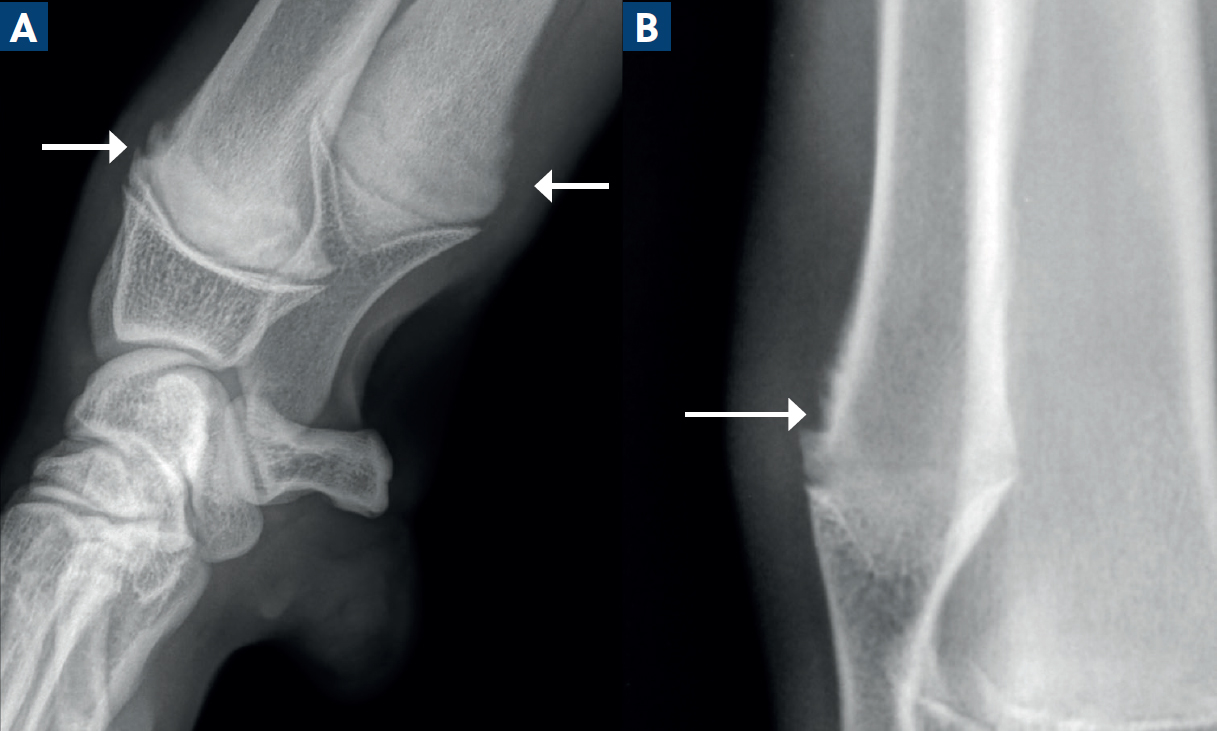
Progression of radiographic changes
Some conditions may not be confidently demonstrated on radiographs until after a period of time; eg bone loss may take 7–10 days to be visible radiologically (Dennis et al, 2010a), and 30–60% of mineral content must be lost before it is detectable.
Follow-up radiographs are useful for reassessing a patient whose initial radiographs were normal, but has not improved clinically, to assess for changes that may have not been visible on the first set of images. Follow-up radiographs may be performed 7–10 days later for hairline fractures, or 3 weeks later for cases of physeal trauma (Dennis, 2010a). A major pitfall of radiographs is the inability to provide any information about damage to articular cartilage or soft tissue structures (Dennis, 2010a).
Classification of lameness
Table 1 is a non-exhaustive summary on the general conditions seen in canine lameness, categorised according to how useful the author found them and largely adapted from Cook (2001). The predisposed breeds are indicated, but mixed breeds are also affected by many of the same disorders.
| Category | Specific conditions or disorders | Breeds* |
|---|---|---|
| Congenital – truly present at birth | Luxation – shoulder | CHI, PO, TP |
| Luxation – elbow | BH, BT, CHI, EBD, GSD, PU, PEK | |
| Dysostoses – extensive group of disorders of abnormal development of individual bones or parts of bones, including ectrodactyly and hemimelia | None | |
| Osteochondrodysplasia | Large and Giant breeds, LAB, BC | |
| Osteogenesis imperfecta | None | |
| Mucopolysaccharidosis | MIN | |
| Developmental – occur during the growth phase | Osteochondrosis/osteochondritis dissecans – disturbance of endochondral ossification | |
|
|
Many breed-specific types; BUT, BO, BE, LAB, TP | |
|
|
GRT, LAB, GSD | |
|
|
Large and Giant breeds | |
|
|
Large and Giant breeds | |
| ROT, LAB, BT, BB†, SBT | ||
| ROT, GRT | ||
| Elbow dysplasia
|
Large and Giant breeds, GSD, LAB, ROT, BMD | |
| Large and Giant breeds, GD, BB, GSD | ||
| None | ||
| Hip dysplasia | Large and Giant breeds eg GSD, LAB, BB, GRT | |
| Retained endochondral cartilage cores | Large and Giant breeds, GD, IWH | |
| Humeral intra-condylar fissures | FBD, PU, SP | |
| Avascular necrosis of the femoral head (Legg Calve Perthes Disease) | Small/miniature breeds | |
| Osteochondroma, multiple cartilaginous exostoses | None | |
| Slipped capital femoral epiphysis – part of osteochondrosis | LAB, AMB† | |
| Medial patellar luxation | Small and Miniature Breeds | |
| Sesamoid disease | ROT | |
| Neoplasia | Lymphoma, osteosarcoma | Any |
| Idiopathic | Panosteitis | GSD, LAB†, BH |
| Hypertrophic osteodystrophy | W, GD, IR, RR† | |
| Infectious | Septic arthritis | None |
| Septic metaphysitis | None | |
| Osteomyelitis | None | |
| Inflammatory | Immune mediated polyarthropathy | None |
| Trauma | Salter Harris Classification for physeal fractures | None |
| Tibial crest avulsion fractures | STB (Gower et al, 2008) |
Breeds that the condition is most seen in, list not exhaustive.
American Bully = AMB; BB = Boerboel; BC = Border Collie; BE = Beagle; BH = Basset Hound; BMD = Bernese Mountain Dog; BO = Boxer; BT = Boston Terrier; BUT = Bullterrier; CHI = Chihuahua; EBD = English Bulldog; FBD = French Bulldog; GD = Great Dane; GRT = Golden Retriever; GS = Giant Schnauzer; GSD = German Shepherd; IR = Irish Setter; IWH = Irish Wolfhound; LAB = Labrador Retriever; MIN = Miniature Pinscher; PEK = Pekingese; PU = Pug; PO = Pomeranian; RR = Rhodesian Ridgeback; ROT = Rottweiler; SP = Spaniel types; STB = Staffordshire Bullterrier; TP (miniature) = Toy Poodle; YT = Yorkshire Terrier; W = Weimaraner †In the author's experience.
Specific conditions
Hip dysplasia
While hip dysplasia does not present to the clinician as an emergency diagnosis, it is important that they are aware that there are certain age limits to specific interventional surgeries, and this must be kept in mind when pelvic limb lameness is as a result of hip dysplasia.
Examples of surgeries include juvenile pubic symphysiodesis, which must be performed between 12 and 18 weeks of age (Linn, 2017), or double or triple pelvic ostectomy. This is usually performed in younger dogs ranging from 5–14 months of age, depending on the presence of arthrosis (Guevara and Franklin, 2017).
Elbow dysplasia (developmental elbow disease)
Elbow dysplasia, or developmental elbow disease, is a polygenic hereditary condition where a genetic test is not available, and thus veterinarians rely heavily on imaging for diagnosis. Three radiographs may be more optimal for full assessment of the elbow, rather than standard orthogonal views, and include the mediolateral neutral view with the elbow at 120°, the flexed mediolateral neutral view (best to assess the anconeus process) with the elbow at 45°, and 15° pronated craniocaudal oblique view (best to assess the medial aspect of the humeral condyle) (Thrall and Robertson, 2016; Vezzoni and Benjamino, 2021).
Elbow dysplasia historically consists of four underlying conditions according to the International Elbow Working Group (Kähn et al, 2023): fragmented medial coronoid process, osteochondrosis/osteochondritis dissecans of the medial humeral condyle, ununited anconeal process and elbow joint incongruity.
A potentially helpful development in radiological screening for underlying medial coronoid process disease is the presence of infra- or subtrochlear sclerosis of the ulna in the region of the abnormal medial coronoid process (Vezzoni and Benjamino, 2021). An increase in medullary opacity is proposed to occur with loss of the normal bony trabecular pattern because of joint inflammation and/or overloading, although one study suggested that this only develops in advanced cases of medial coronoid process disease (Lau et al, 2015) and may be affected by poor observer specificity (Burton et al, 2008). Additional abnormalities include a blunted, irregular, indistinct or less opaque medial coronoid process. Computed tomography remains the gold standard imaging modality for assessing the elbow.
Arthrosis is an important indicator of underlying elbow dysplasia in a young dog, even if the primary lesion is not identified. The typically affected sites that should be carefully scrutinised are the anconeal process, the cranioproximal radial head and lateral humeral condyle (lateral epicondylar crest) (Dennis, 2010b; Vezzoni and Benjamino, 2021) (Figure 2).
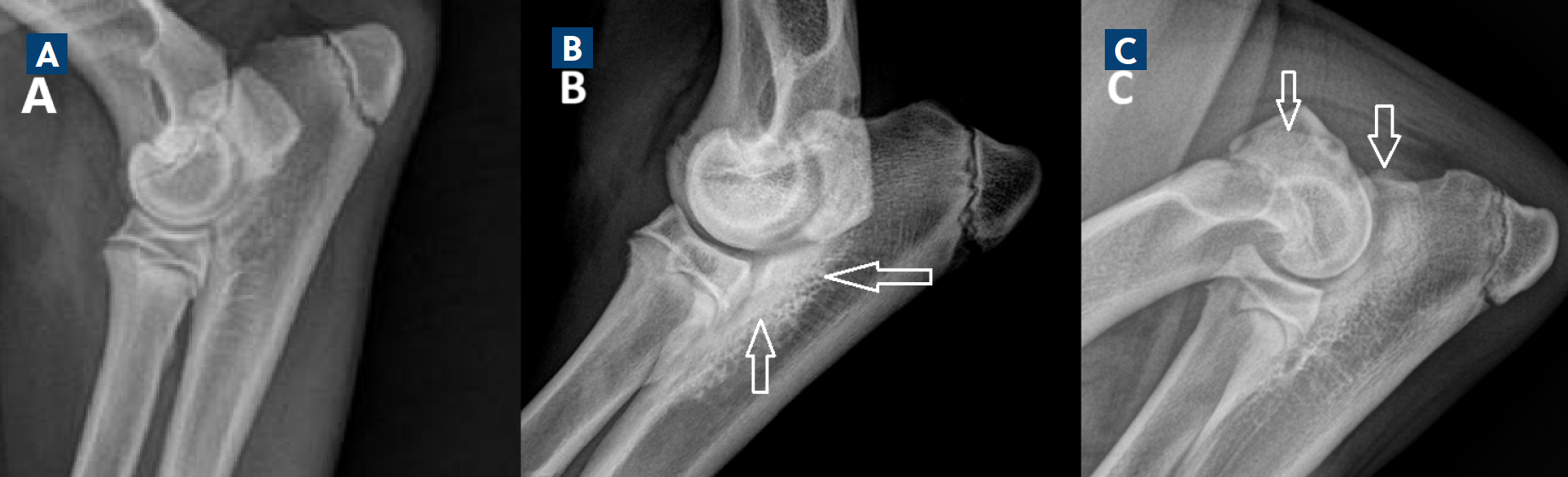
Ununited anconeus process is the presence of a separate anconeus fragment after the accepted date of fusion (approximately 5 months of age). In most breeds, including German Shephard dogs, the anconeal process should be fused by 4 months of age, and in giant breeds, including Great Danes and Saint Bernards, fusion occurs between 4–6 months of age (Vezzoni and Benjamino, 2021).
Medial humeral osteochondrosis/osteochondritis dissecans lesions are best assessed on the pronated craniocaudal image for the typical saucer-shaped defect of the medial humeral subchondral bone. Osteochondrosis is detailed later in this article.
The exact mechanisms of elbow incongruity remain unclear and incongruity, whether radioulnar or humeroulnar, may be challenging to assess on radiographs unless overt. Accurate assessment is dependent on limb positioning, eg degree of flexion, supination or pronation, and loading (weight-bearing) (Alves-Pimenta et al, 2019).
The ideal intervention for elbow dysplasia relies on the veterinarian's identification of, sometimes very subtle, radiological changes and clinical evaluation early on; 4 months of age in medium to large breeds and at 5 months of age in giant breeds) (Vezzoni and Benjamino, 2021), and even earlier than this. Early surgical intervention is possible but is beyond the scope of this article.
Humeral intracondylar fissures
This condition has received increased attention given the increased popularity of the French Bulldog, one of the most affected breeds, as highlighted in the literature (Strohmeier and Harris, 2021; Schettler et al, 2022).
Humeral intracondylar fissure, previously referred to as incomplete ossification of the humeral condyle is a descriptive term for a radiolucent fissure that occurs in between the lateral and medial parts of the humeral condyle (Moores, 2021). The condyle has two centres of ossification, separated by a thin cartilaginous plate that typically fuses and unites them at 8–12 weeks of age (70±14 days); however, the breeds in which this timing occurs is not stated (Hutchings and Rutherford, 2024).
It is suggested that humeral intracondylar fissures may be a developmental condition seen in immature brachycephalic breeds, primarily the French Bulldog and Pug, representing an incomplete or delayed fusion of these centres (Hutchings and Rutherford, 2024) (Figure 3). Given the complex nature of the elbow, multiple radiographs at different angles may be required for diagnosis of this particular condition, especially the 15° craniomedial-caudolateral oblique (supinated) radiograph. Superimposition of the ulna over the humeral condyle on the cranio-caudal view, with resultant Mach lines, can complicate the diagnosis (Strohmeier and Harris, 2021). Computed tomography is the gold standard imaging modality.
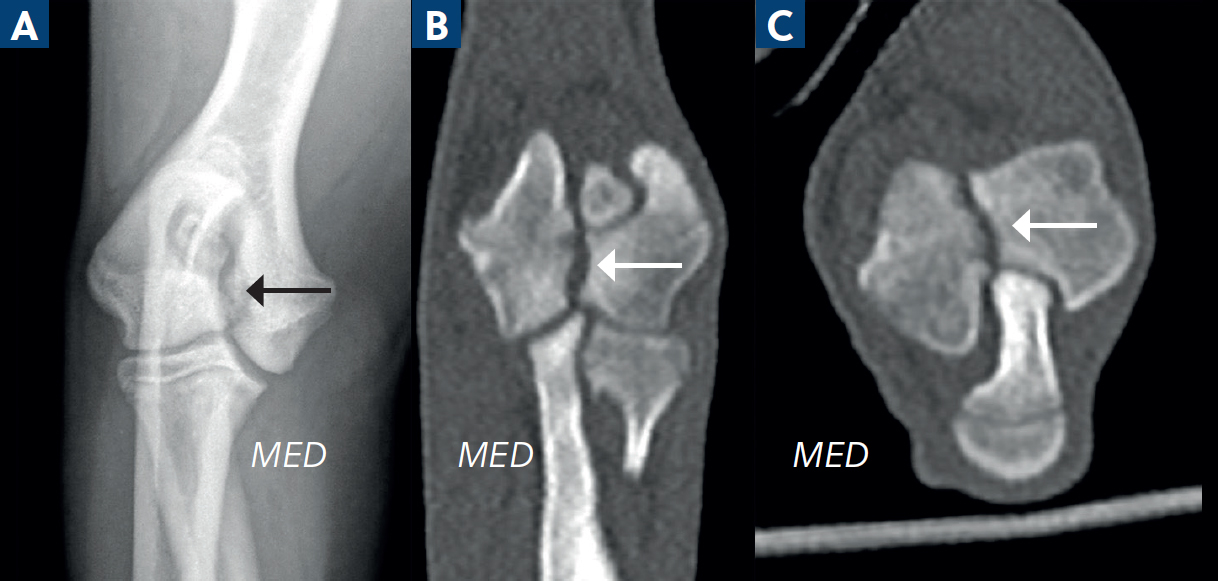
Humeral intracondylar fissures may be clinically silent or result in lameness and pain; the verdict on whether this condition may predispose patients to lateral or medial humeral condylar fractures is up for debate (Anderson et al, 2023). Moores and Moores (2017) reported that 18% of incidental humeral intracondylar fissures went on to fracture within 2 years of diagnosis, but this was in a group over-represented by Spaniels. Another reported that French Bulldogs are 49x more likely to develop a humeral condylar fracture when compared with the general population (Schettler et al, 2022). Humeral intracondylar fissures may often be bilateral (Anderson et al, 2023).
The spontaneous resolution of humeral intracondylar fissures diagnosed in immature French Bulldogs have been noted on follow-up imaging, after the cut-off date of normal ossification had occurred (Garland et al, 2023; Hutchings and Rutherford, 2024). Surgical treatment is outside the scope of this article.
Osteochondrosis and osteochondritis dissecans
Osteochondrosis is a common multifactorial developmental disorder of joints (Ytrehus et al, 2007) and articular cartilage, characterised by a focal disturbance of endochondral ossification. It occurs because of a failure of blood supply to the epiphyseal growth cartilage, and happens in certain joints. In dogs, this is most commonly the shoulder joint, followed by the elbow, tarsal, and stifle joints (Johnson et al, 1994). The term ‘osteochondrosis dissecans’ is used if the cartilage over the lesion fissures off and creates a flap (Dennis et al, 2010a; Engdahl et al, 2024).
Careful inspection of the following regions (Dennis et al, 2010b) for a zone of subchondral bone flattening or a saucer shaped defect, and associated subchondral sclerosis, or a mineralised flap associated with the defect, is recommended once the abnormal joint has been clinically identified and radiographed (Figure 4):
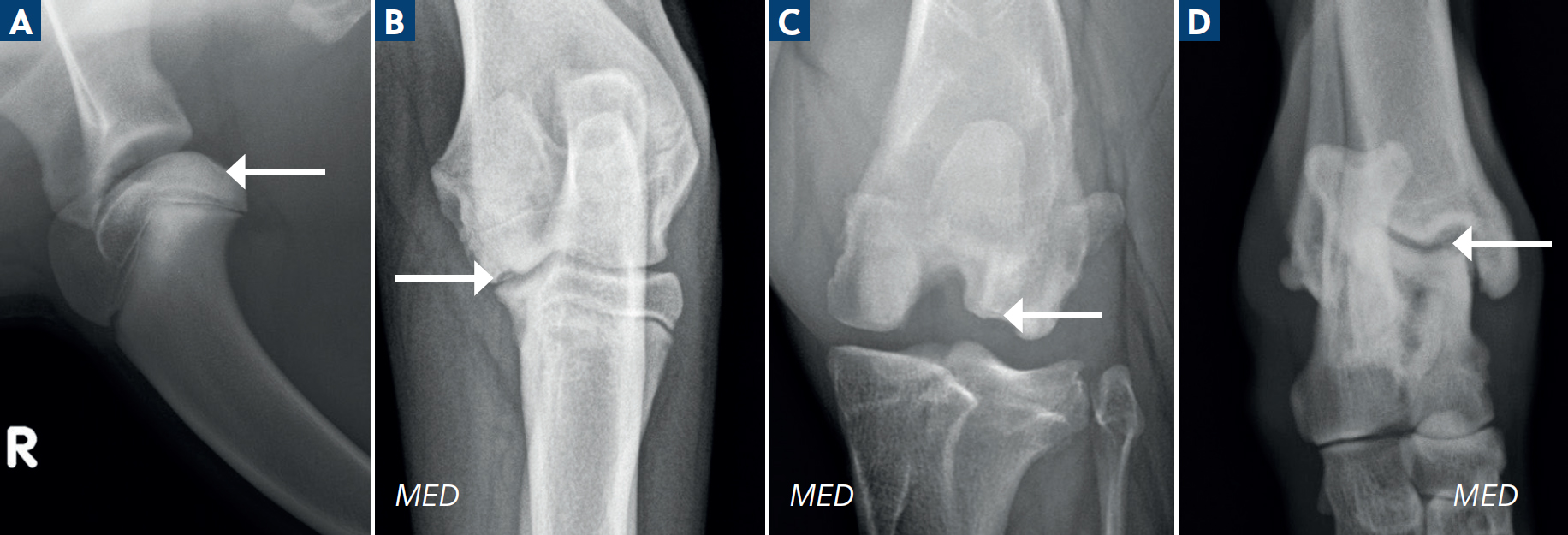
Lateral trochlear ridge lesions are often large fragments, and their identification may be hampered as the calcaneus can superimpose over this region on the standard dorsoplantar radiograph. Several additional projections (Gielen et al, 2005) have been recommended, including the fully extended and flexed mediolateral, the plantarodorsal, the plantaromedial-dorsolateral and plantarolateral-dorsomedial, and flexed dorsoplantar skyline views, given the complexity of this joint (Figure 5). As these conditions may present bilaterally, both joints in any case of osteochondrosis or osteochondritis dissecans should be evaluated with radiographs.
Retained endochondral cartilage cores
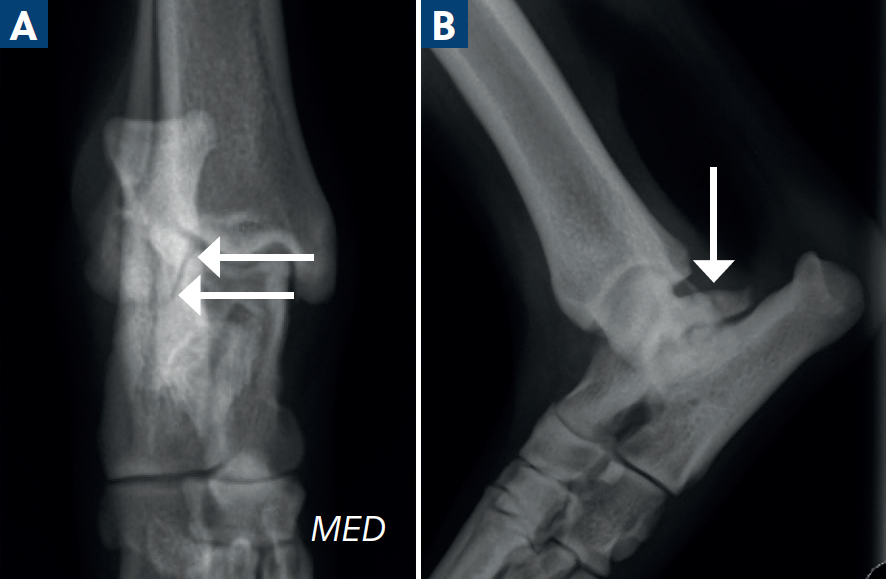
Typically affecting the distal ulna, this condition is represented by zones of hypertrophic chondrocytes (growth plate cartilage), projecting into the associated metaphyses from the physis (Kieves, 2021). They have a radiolucent ‘flame’ or ‘cone’ shape, often surrounded by a fine sclerotic rim. These occur when cartilage fails to convert into normal bone and may be considered a form of osteochondrosis. In some cases, they may resolve spontaneously.
Often, they are clinically silent. However, the importance to the veterinarian is to monitor the patient for any signs of ensuing disruption of the physis, especially in very immature animals that still have significant growth potential before maturity. Disruption can lead to decreased longitudinal growth resulting in angular limb deformities (Figure 6).
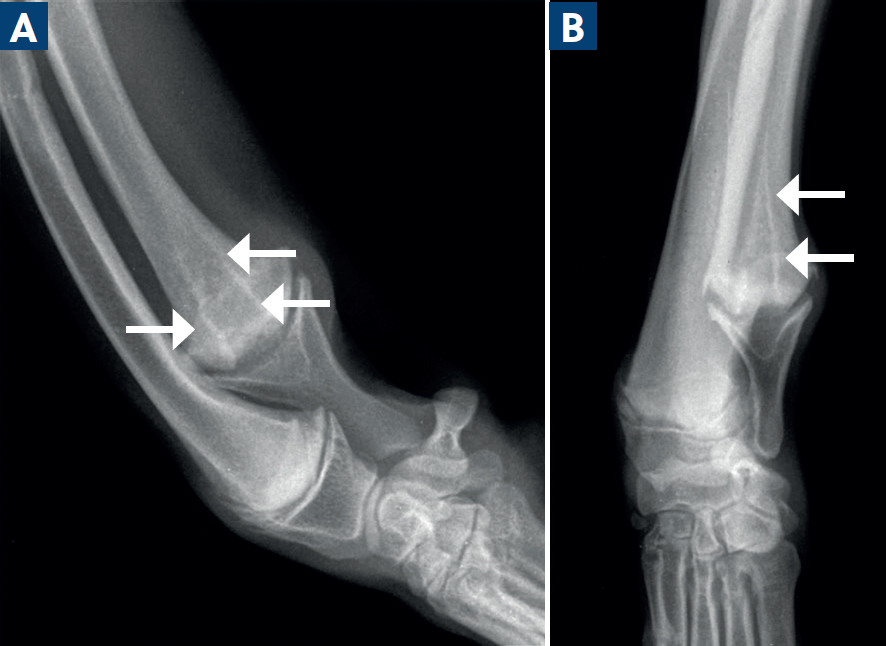
Hypertrophic osteodystrophy
Also known as metaphyseal osteopathy, the underlying aetiology of hypertrophic osteodystrophy remains unknown (Kieves, 2021). Heritable, auto-immune, diet and infectious aetiologies have all been considered. Patients present with swelling of the metaphyses, most often bilaterally. The signs of the condition can vary widely, including lameness, pain, pyrexia, anorexia and less common clinical signs such as abnormal respiratory sounds, oculonasal discharge, haematochezia, diarrhoea, vomiting and even systemic inflammatory response syndrome (Selman and Towle Millard, 2022).
Radiological changes consist of a radiolucent zone paralleling within the metaphysis, called a ‘double physis’ (Selman and Towle Millard, 2022). In some cases, a paracortical cuff of mineralisation can develop along the metaphysis. Multiple bones are usually affected. Clinical signs may precede radiological changes by up to 72 hours, so follow up radiographs are helpful in these cases (Figure 7).
Secondary angular limb deformities may develop if there is disruption of the growth plate; this occurs where new bone forms at the metaphysis and bridges the physis. This is an important complication for the veterinarian to monitor for, even if uncommon.
Physeal injuries
Trauma is a common cause of disturbance to the physes (Stander and Cassel, 2016). The physis is composed predominantly of cartilage which is radiolucent (Dennis, 2010a), making assessment difficult if there are subtle changes.
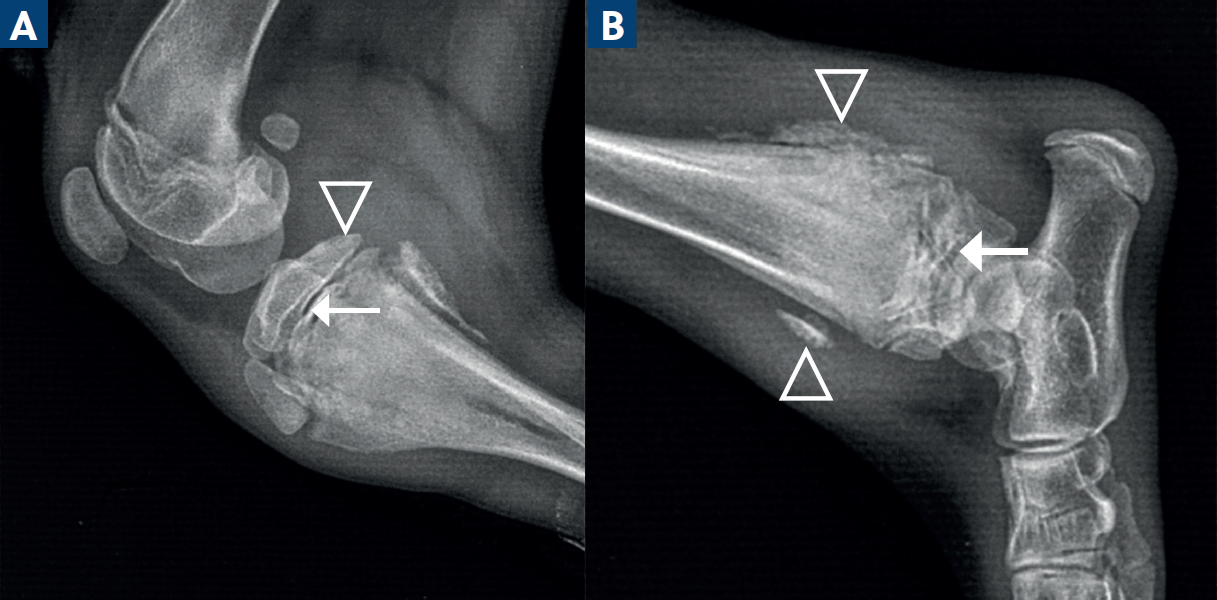
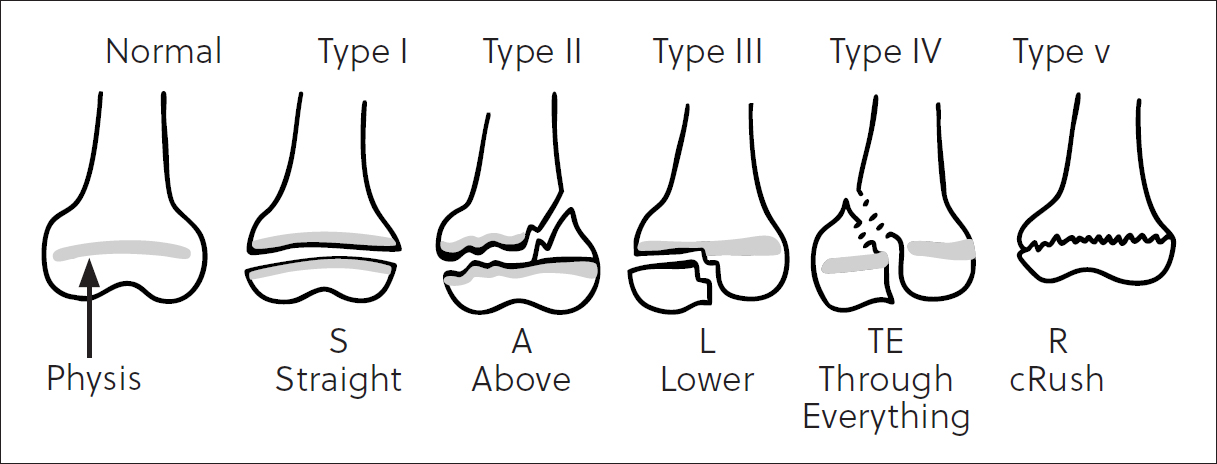
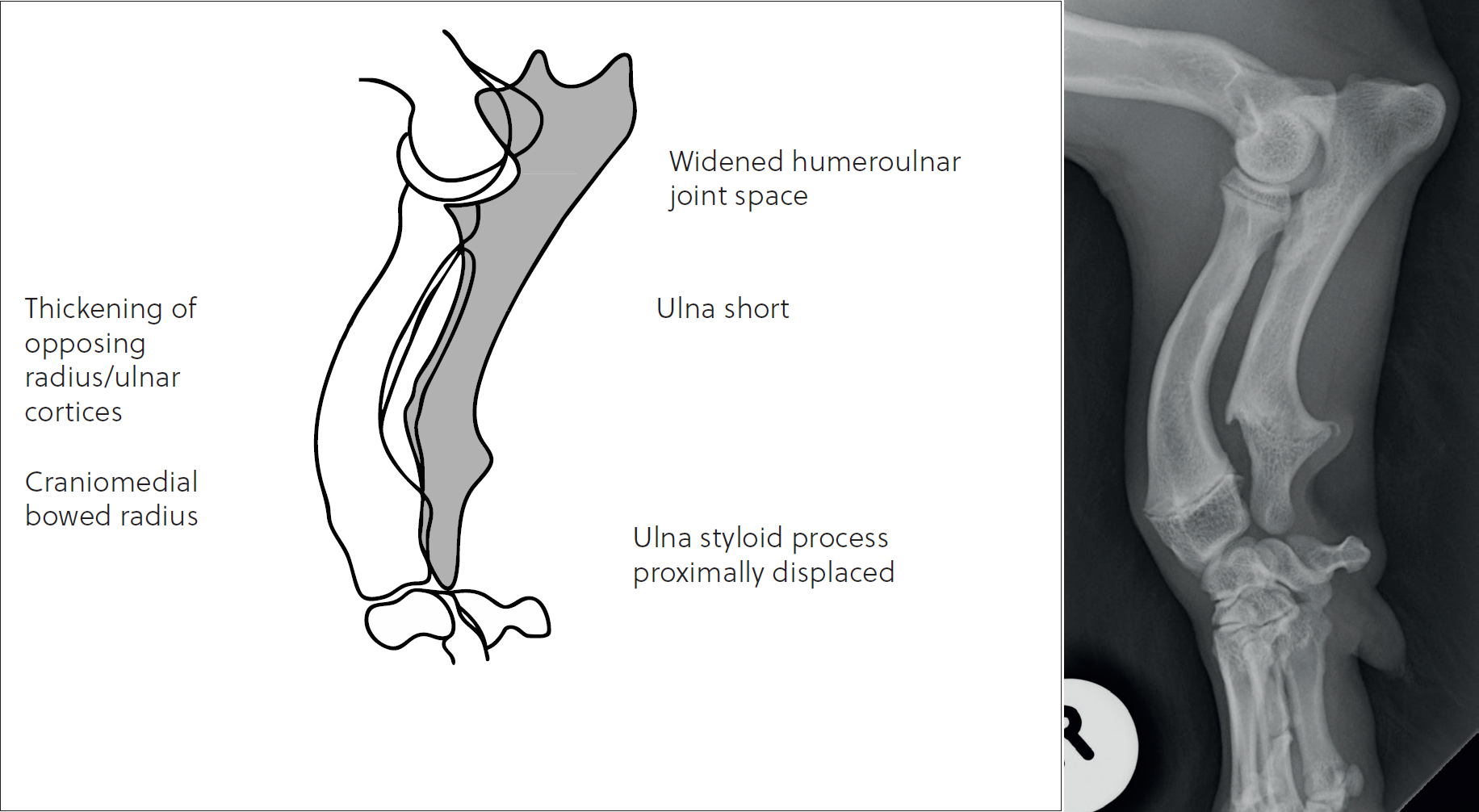
If physeal trauma results in partial or complete premature closure of the growth plate, an angular limb deformity may develop. The severity of deformity depends on the age of the patient at the time of injury – the younger the patient, the greater the growth potential of the physis and the more severe the potential outcome. The severity also depends on which specific physis is affected. Not all physes contribute the same degree of longitudinal growth; eg the longitudinal growth of the ulna relies on 85% (Stander and Cassel, 2016) to 100% (Fox, 2021) originating from the distal physis. Disturbances of relative growth in paired bones where the distal ulna is most affected (Dennis et al, 2010a; Stander and Cassel, 2016; Fox, 2021) result in especially noticeable effects. The Salter Harris classification system is in widespread use to describe physeal fractures (Dennis, 2010a) (Figure 8). While there are in depth reviews on the topic of physeal trauma, and only a few key points are discussed here.
The distal ulnar physis has a unique shape in the canine, described as a ‘cone within a cone’ (Fox, 2021) making it prone to a compression, or Salter Harris type V fracture. It results in a predictable set of changes with regards to the development of the antebrachium as depicted in Figure 9. Subluxation of the elbow (Fox, 2021) (ie humeroulnar incongruity with elongation of the ulnar trochlear notch) can occur concurrently in up to 12% of cases, and thus evaluation of the elbow joint is important.
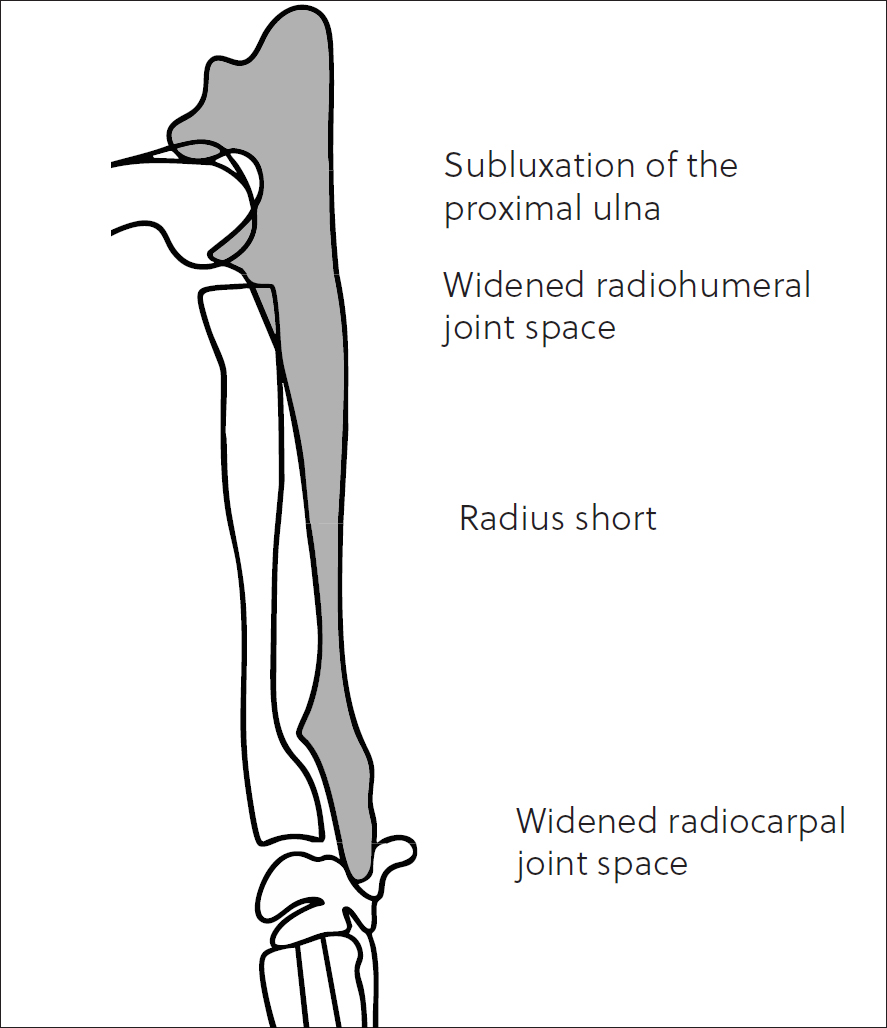
The distal radial physis contributes most of the radial longitudinal growth: 64–70% (Stander and Cassel, 2016; Fox, 2021). Traumatic premature closure can be symmetrical or asymmetrical, and located at the medial or lateral aspect of the physis (lateral appears to be more common). The typical changes are illustrated in Figure 10, assuming symmetrical closure. In cases of asymmetrical lateral closure, the limb can appear as though affected by premature closure of the distal ulnar physis on clinical examination (valgus angulation of the distal radius and carpus). Careful radiological inspection will help distinguish the two; the lateral aspect of the distal radial physis will be narrowed, with lateral deviation of the distal radial epiphysis, resulting in the valgus deformation of the carpus. The ulna may demonstrate recurvatum (caudal bowing) as the radius shortens. Additionally, there may be widening of the humeroradial joint space and humeroulnar subluxation. Early surgical referral and consultation should be discussed with the owners, but treatment is outside the scope of this article.
Conclusions
Radiology remains an affordable and accessible tool for lameness evaluation in the young canine patient. Veterinarians should be pro-active with early imaging in juvenile patients presenting with orthopaedic complaints, rather than adopting a ‘wait-and-see’ approach, which may be more appropriate in adult animals.


