Although diabetes mellitus is a chronic and treatable disease, it is often associated with relatively short survival times in dogs and cats (Niessen et al, 2017; Tardo et al, 2019; Rothlin‐Zachrisson et al, 2023). Approximately 1 in 10 diabetic dogs and cats are euthanised at the time of diagnosis, and an additional 1 in 10 within the first year of therapy (Niessen et al, 2017). Treatment failures commonly arise from temporary or more permanent compliance challenges and frustrations by pet owners. Specifically, the necessity for sustaining a consistent schedule over years can have a negative impact on an owner's quality of life, especially in patients with inconsistent appetite. Pet owners can experience many obstacles to management of their pet's diabetes, such as lifestyle adjustments, unrealistic expectations, financial burden, concerns about potential complications and inability to administer insulin injections. The financial and time commitments can become overwhelming. Frequent veterinary monitoring of glycaemic control and high insulin cost in some geographic locations contribute to these stresses (Niessen et al, 2017). However, there are simple and practical solutions to long-term compliance issues with regards to therapy that can, and should, be implemented by the veterinary practitioner. These require an understanding of diabetes-related pathophysiology, pharmacology and clinical pathology, understanding of general goals of treatments and, critically, a thorough discussion with pet owners about their goals and abilities.
Goals of treatment
The primary goals of diabetes therapy in small animals are:

In patients with residual beta cell function (most cats, almost no dogs) (Ackerman et al, 2018; Shiel and Mooney, 2022), diabetic remission might also be a treatment goal. Achieving specific glycaemic targets is not a primary goal, but rather serves as a guide for treatment (Niessen et al, 2022). It is impossible to tighten glycaemic control with insulin therapy without increasing the risk of hypoglycaemia. It is rarely the case that tight glycaemic control is required for prevention of diabetic ketoacidosis (Behrend et al, 2018; Restine et al, 2019; Gilor and Fleeman, 2022). Moreover, an effort to achieve specific glycaemic targets might negatively affect the owner's quality of life and lead to unnecessary expenses. Realistic goals should be determined after thorough discussion with the pet owner on the feasibility of implementing potential treatments and monitoring strategies. It is important to keep in mind that the expectations of pet owners are often set by the veterinarian's expectations. Therefore, a discussion on treatment goals and the strategies to implement them should be done with self-awareness and sensitivity to the owner's needs and abilities.
Communication is vital to determine an owner's priorities, and treatment should be customised to the individual patient and owner. Compromise to determine the ‘best’ therapy for both the pet owner and individual pet is key. The ‘ideal’ therapy for successful diabetes treatment would have high owner and pet compliance, be less expensive and rapidly effective, have minimal risk of hypoglycaemia and decreased impact on owner's quality of life.
What is the ‘best’ insulin?
In veterinary medicine, there are no studies showing a specific benefit of one insulin formulation over another, in any clinical context. Therefore, there is no insulin which is the ‘best’ for diabetic dogs and cats. Instead, the insulin choice should be personalised to the individual pet and client. Insulin choice is dependent on many factors such as costs, availability, concurrent diseases, owner adherence, monitoring strategies, diet and feeding regimen and specific goals of diabetes therapy (Fleeman and Gilor, 2023). There are a range of insulin formulations available in the human market, but only two insulin formulations approved for veterinary use – Prozinc and Vetsulin (Caninsulin in Europe) – for the treatment of diabetes in dogs and cats (Table 1). In certain countries, such as the United Kingdom, diabetic dogs and cats must be treated first with an approved veterinary insulin before other insulins can be used (Ackerman et al, 2018), while in other countries like the USA, off-label use of drugs approved for humans is permitted and often preferred.
| Brand name | Formulation | Concentration (units/ml) | Vial or pen | Frequency | Basal or bolus | |||
|---|---|---|---|---|---|---|---|---|
| Canine | Feline | Canine | Feline | |||||
| Tresiba | Human recombinant insulin degludec | 100/200 | Pen | Solution | Every 12–24 hours | Every 12 hours | Basal | Bolus |
| Toujeo | Human recombinant insulin glargine | 300 | Pen | Solution | Every 12–24 hours | Every 12–24 hours | Basal | Basal |
| Lantus | Human recombinant insulin glargine | 100 | Vial and pen | Solution | Every 12 hours | Every 12–24 hours | Basal | Bolus |
| Prozinc* | Human recombinant protamine zinc insulin | 40 | Vial | Suspension | Every 12–24 hours | Every 12–24 hours | Bolus | Bolus |
| Vetsulin* Caninsulin* | Porcine lente | 40 | Vial and pen | Suspension | Every 8–12 hours | Every 8–12 hours | Bolus | Bolus |
| Humulin N Novolin N | Human recombinant neutral protamine Hagedorn | 100 | Vial and pen | Suspension | Every 8–12 hours | Bolus | Bolus | |
Veterinary approved
Insulin suspensions have greater day-to-day and intra-day variability (Heise et al, 2004; Miller et al, 2021; Fleeman and Gilor, 2023), and they must be mixed (shaken vigorously in the case of Vetsulin/Caninsulin) before administration. Inconsistent dosing (resulting from inconsistent resuspension) and variation in absorption from the subcutaneous depot are significant contributors to between-day variation of insulin suspensions. The unpredictable time-action profile of these suspensions hinders the ability to quickly and safely escalate insulin doses, minimise hypoglycaemia and requires close monitoring (Gilor and Fleeman, 2022).
Generally, solutions (such as glargine, detemir or degludec) are more predictable with less day-to-day variations compared to suspensions (neutral protamine Hagedorn, Lente, protamine zinc insulin) (Heinemann et al, 2000; Heise et al, 2004; Sako et al, 2011; Oda et al, 2018; Miller et al, 2021). Solutions do not require resuspension and mixing, making dosing more accurate. Absorption of insulin solutions from the subcutaneous depot is more predictable. Insulin solutions such as glargine and degludec are usually less potent and have a longer duration of action than suspensions (Gilor and Fleeman, 2022; Fleeman and Gilor, 2023). Many insulin formulations come in both injection pens and vial/syringe forms (Table 1). When possible, injection pens are preferred over vials and syringes.
Insulin pens
There are several advantages to using an insulin injection pen in comparison to insulin syringes and vials. Notably, insulin pens increase accuracy and precision, particularly for lower dosages (<5 U) (Gnanalingham et al, 1998; Keith et al, 2004; Luijf and DeVries, 2010). Even when administered by experienced veterinarians and technicians, imprecision of >20% occurs approximately 50% of the time when using an insulin syringe to draw 1 U (Borin-Crivellenti et al, 2014). The adoption of insulin injection pens increased treatment adherence, reduced hypoglycaemic events and improved owner satisfaction and patient quality of life (Cobden et al, 2007; Pfützner, 2009; Asche et al, 2010; Dang and Lee, 2010). The VetPen (Merck Animal Health), which contains 40 IU/ml of porcine zinc suspension, is the only approved injection pen for veterinary use. Two versions exist: 8 units/pen with 0.5 U increments and 16 units/pen with 1 U increments. When compared to insulin syringes, the VetPen is more accurate at very low doses (1 U), while syringes tend to surpass the intended dose (Burgaud et al, 2012). There are also many non-veterinary approved pens, including glargine U-100, Novolin N U-100, Toujeo U-300 and degludec U-100/U-200.
In some regions, the cost of injection pens can be a limiting factor, especially if insulin is discarded according to recommended manufacturer expiration dates (McQueen and Li, 2023; Pendsey et al, 2023). Injection pen cost can be reduced in several ways. First, the pen should be primed with 1–2 units (air shot) before the initial injection, but does not necessarily need to be primed before each subsequent injection in the author's experience (as is recommended in humans) (Gilor and Fleeman, 2022). This eliminates unnecessary insulin wastage. Air-shots should be performed if there are bubbles in the reservoir or variability in insulin predictability. The insulin pen should be tilted with the needle pointing down during injection to avoid administration of bubbles that may decrease the intended dose. Second, insulin pens do not need to be discarded until empty, or until there is suspected contamination or ineffectiveness. Insulin is stable in solution, and when refrigerated, Eli Lilly cites a linear decrease of <0.1% in activity per month (Grajower et al, 2003). To save money and maintain efficacy, insulin pens should be refrigerated between use and allowed to warm to room temperature (10–15 minutes) before administration. Contamination is also very rare, with one study demonstrating that refrigerated insulin vials containing standard preservatives (such as phenol or metacresol) did not show microbial contamination over 6 months of twice daily use (Berg et al, 2023).
Using insulin injection pens is easier with 8–12.7 mm needle tips in veterinary patients, in contrast to 4 mm tips that are preferred in humans. The authors observed that 4 mm needle tips often bend during administration of insulin in dogs and cats. Additionally, it is the authors’ opinion that most veterinary patients do not need to have the needle remain in the skin for 5 seconds unless administering large doses (>20 U). Once the dial returns to zero, the pen can be removed.
Insulin cost
The cost of insulin varies between countries. In the USA, most insulin formulations are relatively expensive, and the long-term expense can easily become a critical factor in the owner's decision on whether to treat. At the time of writing, Walmart brand (Novolin N and Novolin 70/30) insulin and insulin degludec were the most cost-effective insulins for treating diabetes in dogs in the USA. However, these formulations are not recommended for cats. For cats, the most cost-effective formulations are the Lantus-biosimilars: Semglee and Basaglar.
The diabetic dog with a finicky appetite
For diabetic dogs with finicky appetites, investigation of possible underlying concomitant diseases, such as pancreatitis or inflammatory bowel disease, is essential. However, in the author's experience, such investigation often fails to identify a treatable disease. Increased pancreatic lipase immunoreactivity consistent with pancreatitis is often measured in diabetic dogs but is not correlated to clinical signs (Hamilton et al, 2021; Kim et al, 2023). ‘Leaky gut’ associated with diabetes in dogs may contribute to the finicky appetite (Jergens et al, 2019; Crakes et al, 2021).
The traditional approach for insulin therapy in dogs that skip meals is to administer half of the normal amount of insulin, as giving the same dose of insulin can cause hypoglycaemia (Figure 2). This assumes that a standard dose of intermediate-acting insulin suspension will be adjusted based on the expectation of peak insulin action, corresponding to peak carbohydrate absorption from the digested meal. This approach is problematic because it can result in over- or under-dosing of insulin and create significant stress for owners. In situations where dogs with diabetes have inconsistent appetites, no identifiable treatable cause and behavioural modification is unsuccessful, two alternative strategies can be effective:
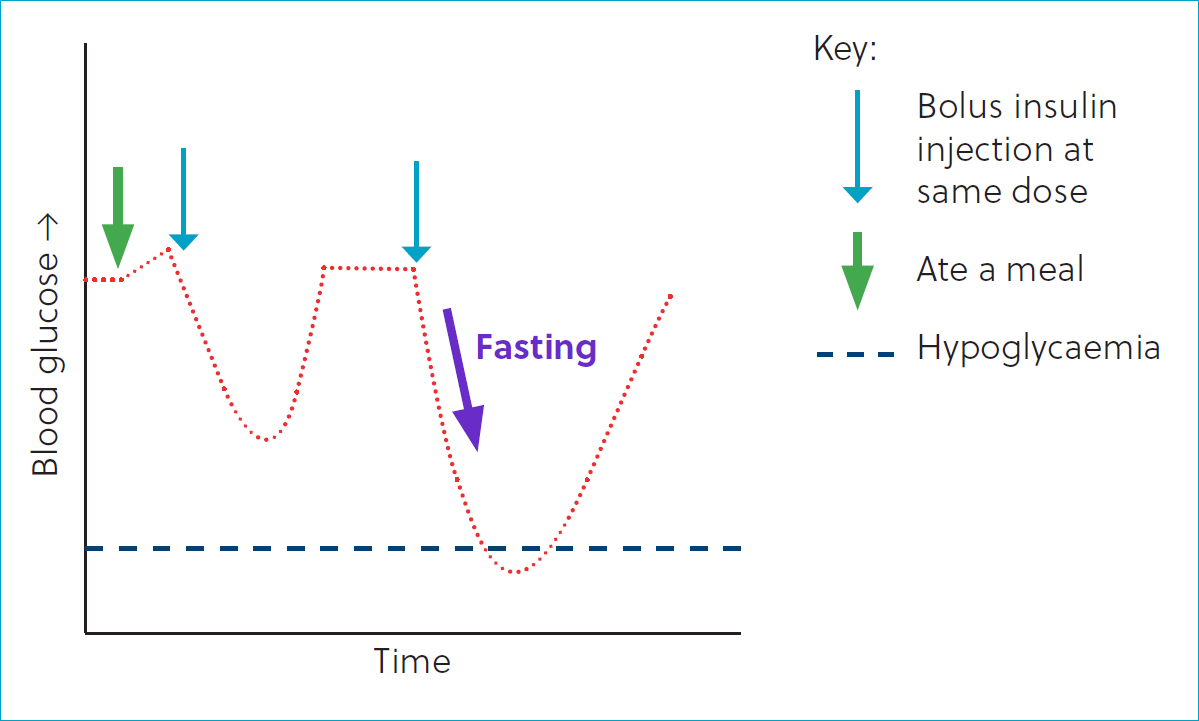
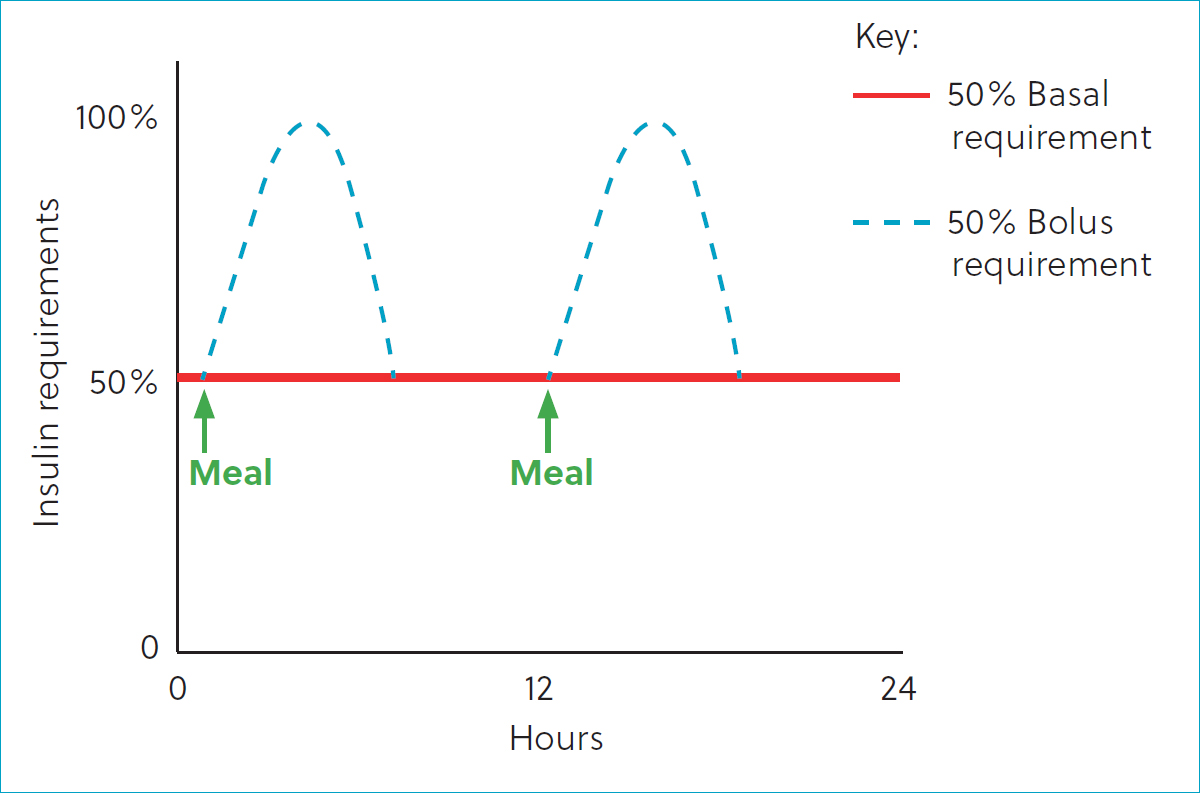
Endogenous insulin is secreted in two phases: basal and bolus. Basal secretion of insulin during fasting is at a consistent rate. In the bolus phase, insulin secretion increases rapidly in response to carbohydrate ingestion and then wanes off rapidly when those carbohydrates have been digested and absorbed (Owens and Bolli, 2008; Gilor and Graves, 2010) (Figure 3). On average, bolus requirements represent about 50% of the daily total insulin requirement (Owens and Bolli, 2008). Novel synthetic insulins are designed to mimic one of the two secretion phases. Humans with Type 1 diabetes often take a daily ‘basal’ insulin and additional ‘bolus’ insulin injections corresponding to their meals, and dogs with diabetes can be treated with a similar basal–bolus combination. However, most diabetic dogs do well clinically with a basal insulin alone (Gilor and Fleeman, 2022; Gilor et al, 2022). This simplified treatment protocol still allows the administration of insulin regardless of food consumption, often with a single injection per day, and alleviates stress for owners who have diabetic dogs with inconsistent appetites. Effective basal insulin therapy typically involves the use of long-acting recombinant insulin analogues, like Toujeo or degludec, which closely mimic the natural pattern of basal insulin secretion (ie a single dose is absorbed at a relatively constant rate throughout a 24-hour period with no significant peaks). Basal insulin doses are adjusted based on continuous glucose monitoring data in the fasting state. A bolus insulin can be added in dogs that do not achieve good glycaemic control on basal insulin alone. Bolus insulin injections are given immediately after meals and the dose is adjusted according to food consumption. On most commercial dog foods and with twice daily feeding, the bolus phase usually lasts between 6–9 hours (Hill et al, 2006; Carciofi et al, 2008; Elliott et al, 2012). Therefore, rapid-acting formulations that are used in humans as bolus insulin and last less than 4 hours are usually insufficient in dogs. Instead, low dose (0.1 U/kg) ‘intermediate’acting formulation (eg neutral protamine Hagedorn, Lente) can be used. Alternatively, dietary adjustments can be made with reduction of carbohydrate content to reduce post-prandial glucose spikes instead of adding bolus insulin.
Using a continuous glucose monitor can help assess insulin requirements over several days of treatment with several different diets. For dogs with finicky appetites, different diets can be offered sequentially, each for an adequate duration (about 3–5 days or as long as needed to establish a pattern of response), while closely monitoring and adjusting insulin doses. Once this process is complete, owners are instructed to administer insulin based on the type and quantity of food the dog is eating. When switching between diets, insulin overdosing can occur when using an insulin with duration exceeding 12 hours. Precautions should be taken, such as observing continuous glucose monitoring data and lowering insulin dose when switching to a lower carbohydrate diet until the effects of the higher insulin dose have resolved.
The challenge of ‘miniature’ dogs and insulin therapy
Managing glycaemic control in patients that are small (eg toy breed dogs or dogs weighing <4 kg) or extremely sensitive to insulin presents significant challenges. Currently, there are no available insulin formulations more dilute than 40 units/ml, and with existing pens and syringes, administrating doses smaller than 0.5 units with precision and accuracy is nearly impossible. The VetPen can deliver Vetsulin in 0.5 unit increments. The Junior QwikPen can also administer 0.5 units, but it delivers insulin lispro, which is too short-acting as a sole insulin for practical use in most dogs and cats (Fleeman and Gilor, 2023). Drawing doses in the 0.5 unit range in insulin syringes (U-100) results in considerable day-to-day variability (Gnanalingham et al, 1998; Keith et al, 2004). Given these limitations, there are no perfect solutions for pets requiring less than 1 unit of insulin. The idea of diluting insulin may be tempting, but it should never be done. For all insulin formulations currently on the market, the time-action profile is dependent on the kinetics of absorption from the subcutaneous depot which, in turn, depends on insulin concentration. Diluting insulin alters its time-action profile, usually accelerating its absorption (Thompson et al, 2015; Saini et al, 2021). Thus, a ‘long-acting’ insulin cannot be assumed to remain long-acting if it is diluted.
In some dogs that require less than 1 unit of insulin, simply switching to 1 unit of long-acting basal insulin can resolve the problem. With basal insulin, such as Toujeo or degludec insulin, 1 unit is slowly delivered over a 24-hour period and peaks are avoided, effectively delivering about 0.5 units of insulin over 12 hours. Another option is to use an insulin pump like the OmniPod. The OmniPod is a tubeless patch pump that requires removal and replacement every 3 days. Its minimum basal flow rate is 0.05 units/hour, equivalent to 0.6 units over a 12-hour period. In the authors’ opinion, this is the safest and most precise method for delivering small insulin doses and yielding the best outcomes, however it may be cost-prohibitive to some owners.
U40 insulin formulations should be administered with U40 syringes. However, if no other option is available, U100 syringes (with 0.5-unit increments) can be used to draw U40 insulin. The volume of a single U40 insulin ‘unit’ is 0.025 ml, and the volume of a single ‘unit’ in a U100 syringe is 0.01 mL. Therefore, by using a U100 syringe with a U40 insulin, one can draw 0.4 U of the U40 insulin with roughly the same precision as drawing a whole unit with a U40 syringe. Drawing 0.5 ‘units’ with a U100 syringe marked at 0.5 U increments would effectively allow drawing 0.2 U of the U40 insulin, albeit with reduced precision (Borin-Crivellenti et al, 2014). If using this strategy, it is crucial to inform all potential caregivers and local clinics about the unconventional use of U40 insulin with the ‘wrong’ syringe.
Dietary therapy in cats with diabetes
In cats consuming low carbohydrate diets, there is often not a need for bolus insulin (Gilor and Fleeman, 2022). Additionally, in cats that graze on dry food through multiple small meals, bolus insulin requirements may not be clinically relevant (Camara et al, 2020; Gilor and Fleeman, 2022). Even if a cat is fed 1 or 2 meals of dry food, using an intermediate-acting insulin to address post-meal hyperglycaemia may not be clinically appropriate, as carbohydrate absorption from the diet can vary significantly in timing post meal. Additionally, many cats still retain some capacity to produce and release insulin, meaning that exogenous insulin is not the sole factor influencing their blood glucose levels (Rothlin‐Zachrisson et al, 2023). In light of these considerations, cats should be treated with long-acting insulin regardless of the timing of their food consumption.
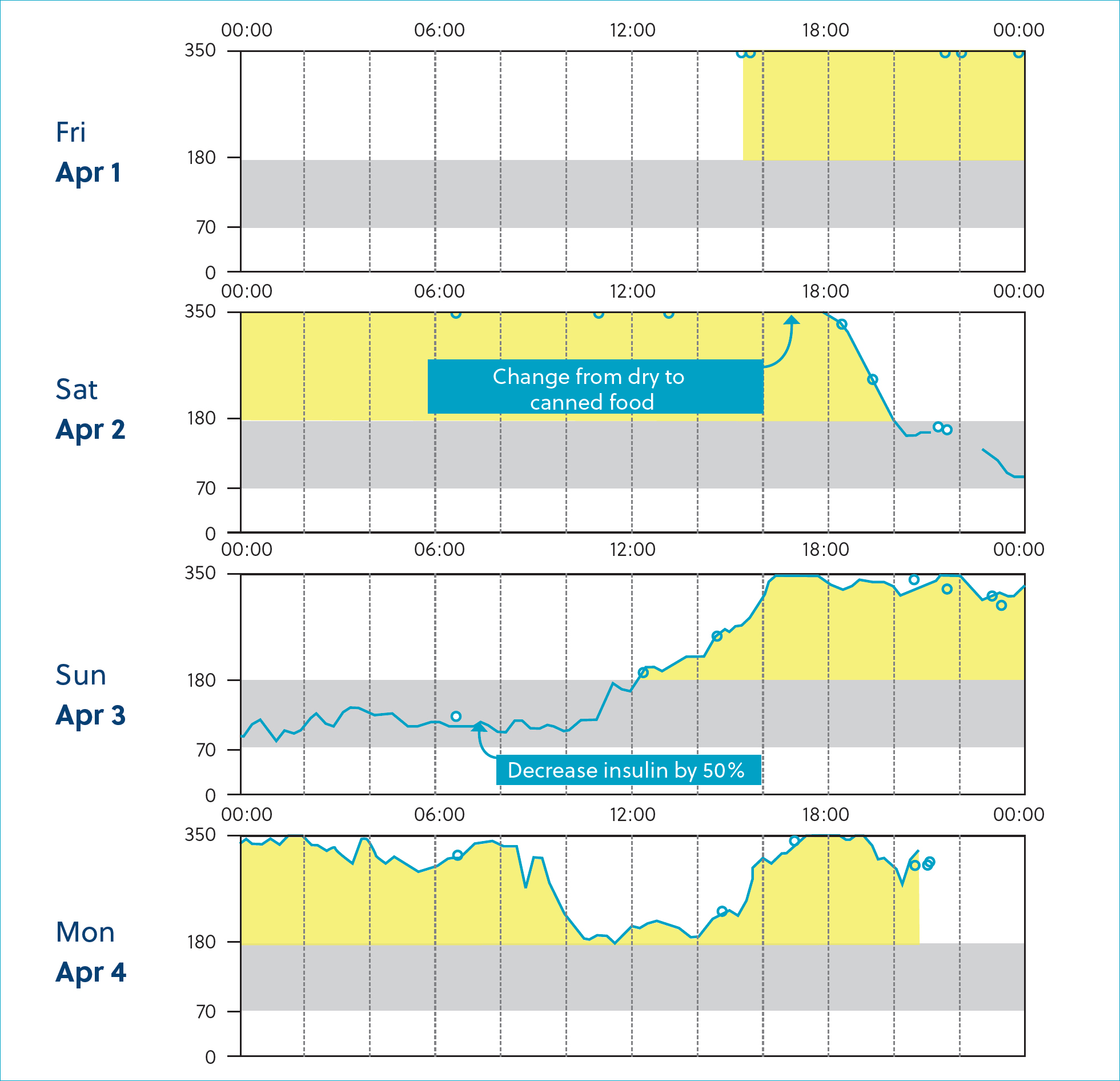
Low carbohydrate (<35 g/Mcal) or ultra-low carbohydrate (15 g/Mcal) diets will reduce insulin requirements and may facilitate achieving diabetic remission (Mazzaferro et al, 2003; Bennett et al, 2006; Gostelow et al, 2014; Parker and Hill, 2023). There are various prescription diets formulated for feline diabetes that feature ultra-low carbohydrate content and are appropriate for therapy. However, if a cat does not find the prescription diet palatable or it is not financially feasible for the pet owner, transitioning the cat from a dry to canned version of the diet will often lead to a reduction in carbohydrate intake and promote weight loss. One study found that cats fed a commercially available wet diet exclusively had increased remission rates compared to those fed either dry or a combination of wet and dry prescription diets for diabetics (Rothlin‐Zachrisson et al, 2023). Figure 4 illustrates the dramatic and acute decline in interstitial glucose levels and the need for an insulin dose reduction in a diabetic cat that was transitioned from a dry, prescription low-carbohydrate diet (carbohydrate content 37 g/Mcal) to an over-the-counter canned diet (carbohydrate content 20 g/Mcal).
Remission in cats
An additional aim in some cats is achieving diabetic remission. Some studies have shown that cats attain remission with tight glycaemic control (Marshall et al, 2009; Roomp and Rand, 2009; 2012), where the aim is to achieve euglycaemia, whereas another study had acceptable remission rates with ‘loose-control’ of diabetes, where the aim is to minimise clinical signs with minimal emphasis on glycaemic values and less risk of hypoglycaemia (Restine et al, 2019). Only one study compared the two approaches side-by-side, and in that study, tight glycaemic control was associated with higher rates of remission compared to loose glycaemic control (Nack and DeClue, 2014). Diabetic remission occurs when endogenous insulin production outweighs endogenous insulin requirement. This usually happens because of a combination of factors such as reducing carbohydrate load (by adjusting diet), improving insulin sensitivity (by resolving concurrent diseases and treating obesity), resolving glucotoxicity (through insulin therapy and in some cases, the addition of glucagon-like peptide 1 analogues) and in some cats, increasing glucose excretion through the kidneys (sodium-glucose cotransporter-2 inhibitors).
As cats with hypersomatotropism and diabetes are unlikely to obtain long-term remission without therapies such as hypophysectomy (Niessen, 2023), tight glycemic control with intensive monitoring and expensive therapy are typically not justified. Cats with diabetes (even those without the typical phenotype) should be screened for hypersomatotropism. Identifying hypersomatotropism early in cats with diabetes allows veterinarians to formulate more appropriate goals and therapeutic plans.
Oral hypoglycaemics: sodium-glucose cotransporter-2 inhibitors
With the release of Bexacat (bexagliflozin) and Senvelglo (velagliflozin) in some countries, there are additional options for treatment of feline diabetes that may be appealing to owners that cannot give injections. Sodium-glucose cotransporter-2 inhibitors function by reducing the reabsorption of glucose in the proximal renal tubules (Lee et al, 2007). This leads to increased excretion of glucose and sodium into the urine and reduction of hyperglycaemia. The glucose lowering effect is rapid and is independent of insulin secretion, beta cell function, and insulin sensitivity. For otherwise healthy, newly diagnosed diabetic cats that have not been treated previously with insulin, sodium-glucose cotransporter-2 inhibitors offer an effective oral therapeutic option (Hadd et al, 2023). In fact, sodium-glucose cotransporter-2 inhibitor therapy might be the fastest and safest way to resolving glucotoxicity and recovering beta cell function. However, for safe and effective treatment with sodium-glucose cotransporter-2 inhibitors as a sole therapy, it is essential to choose appropriate candidates without concurrent conditions. Cats treated with sodium-glucose cotransporter-2 inhibitors must be able to produce some endogenous insulin to prevent diabetic ketoacidosis (Hoenig et al, 2018; Hadd et al, 2023). Cats with a history of diabetic ketoacidosis are not good candidates for sodium-glucose cotransporter-2 inhibitor therapy (Hoenig et al, 2018; Gal and Odunayo, 2023; Hadd et al, 2023). Other concurrent diseases will increase the risk of diabetic ketoacidosis (Rudloff, 2017; Gal and Odunayo, 2023; Hadd et al, 2023). Cats with diabetes on sodium-glucose cotransporter-2 inhibitors should have regular monitoring for development of ketosis. Monitoring of blood beta hydroxybutyrate concentrations may give the earliest indication of insulin deficiency and diabetic ketoacidosis.
What this means for the UK practitioner
In the UK, the licensing and use of insulin for managing diabetes mellitus in dogs and cats is regulated by the Veterinary Medicines Directorate, which mandates that only veterinary-approved insulin formulations, such as Vetsulin and Prozinc, should be prescribed. This contrasts with the USA, where veterinarians can use offlabel human insulins more freely. However, the cascade system, under the Veterinary Medicines Regulations, allows veterinarians to prescribe medications for animals when no suitable licensed veterinary product is available on a case-by-case basis. There is no one rule for everyone, it needs to reflect the patient. This becomes relevant when a licensed insulin is ineffective at meeting the goals of treatment. Longer-acting insulins licensed in humans, such as glargine or degludec, can be considered after veterinarians have demonstrated prior failures with insulins licensed for veterinary use. This prioritisation of treatment should be transparently discussed with pet owners, allowing UK veterinarians to provide a tailored approach to treatment that aligns with Veterinary Medicines Directorate guidelines.
Conclusions
Treating diabetes in dogs and cats requires a tailored approach that considers both the needs of the patient and the goals of the owner to improve compliance. By setting realistic goals and customising treatment plans, veterinarians can empower pet owners to successfully manage their pet's diabetes. Whether choosing a synthetic insulin, an insulin approved for veterinary use or a basal or bolus approach, there is no ‘best’ insulin for all dogs and cats with diabetes. Insulin pens can offer a precise and user-friendly option. For diabetic dogs with an inconsistent appetite, dietary adjustments and/or the incorporation of basal insulin administration that can be decoupled from feedings can be effective solutions. Insulin requirements can often be reduced in cats with diabetes by reducing the dietary carbohydrate content. In some cats, sodium-glucose cotransporter-2 inhibitors may be an oral alternative to insulin. Ultimately, individualising diabetic therapy can enhance quality of life for both pets and their owners.


