Immune-mediated haemolytic anaemia is a significant contributor to illness and mortality in dogs (Figure 1) and, while less common, the condition can also affect cats. Immune-mediated haemolytic anaemia is classified as associative when it can be linked to an underlying disease and non-associative when no specific cause can be identified. Addressing the diseases responsible for immune-mediated haemolytic anaemia can potentially reduce or halt immune-mediated destruction of red blood cells, thus avoiding the adverse effects of long-term immunosuppressive treatment. Possible underlying causes include infections, cancer, medications, vaccines and inflammatory processes (Garden et al, 2019).
Diagnosis
There is no universally accepted standard for diagnosing immune-mediated haemolytic anaemia in veterinary medicine (Garden et al, 2019). Therefore, the results of the diagnostic tests mentioned in this article should be considered in conjunction with other diagnostic tests and the patient's response to immunosuppressive treatment.
Classically, immune-mediated haemolytic anaemia presents as a strongly regenerative anaemia; however, it has been noted that 30% of dogs have a non-regenerative anaemia at first presentation (Piek et al, 2008; Swann et al, 2019), which means that a lack of regeneration does not rule out immune-mediated haemolytic anaemia. It can take 3–5 days for a regenerative response to be seen, and acute cases may show pre-regenerative anaemias.
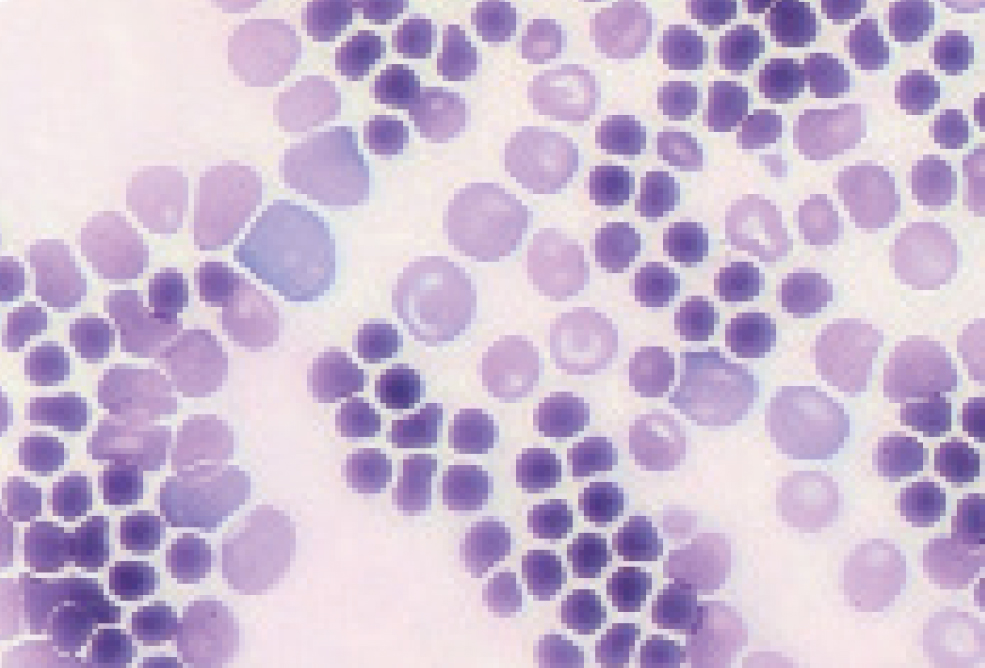
Regeneration is most commonly measured by assessing the reticulocyte count. Reticulocytes are not seen on routine haematoxylin and eosin stains, so new methylene blue stain is required instead (Weiss and Wardrop, 2010). A reticulocyte count can be done in-house by mixing 2 drops of ethylenediaminetetraacetic acid whole blood in a tube containing 2 drops of new methylene blue. The tube should then be inverted and left for 15 minutes at room temperature. A blood smear is then made, and the reticulocytes will have blue staining material (RNA) within the red blood cells. As a result of their higher RNA content, immature anucleated red blood cells can also be recognised as polychromatophilic (purple) red blood cells on Romanowskystained blood smears (Diff-Quik, Wright's stain) and regeneration can be subjectively estimated from the degree of polychromasia in blood smears from both dogs and cats (Swann et al, 2019) (Figure 2).
The reticulocyte count can be measured as follows (Weiss and Wardrop, 2010):
Absolute reticulocyte count (×109/litre) = observed reticulocyte count (%) × total red blood cell count (×1012/litre) × 10
In dogs, an absolute reticulocyte count >60 × 109/litre is consistent with a regenerative anaemia. In cats, an absolute reticulocyte count >50 × 109/litre is consistent with a regenerative anaemia (Swann et al, 2019). Most haematology analysers will provide accurate absolute reticulocyte counts.
Immune-mediated destruction
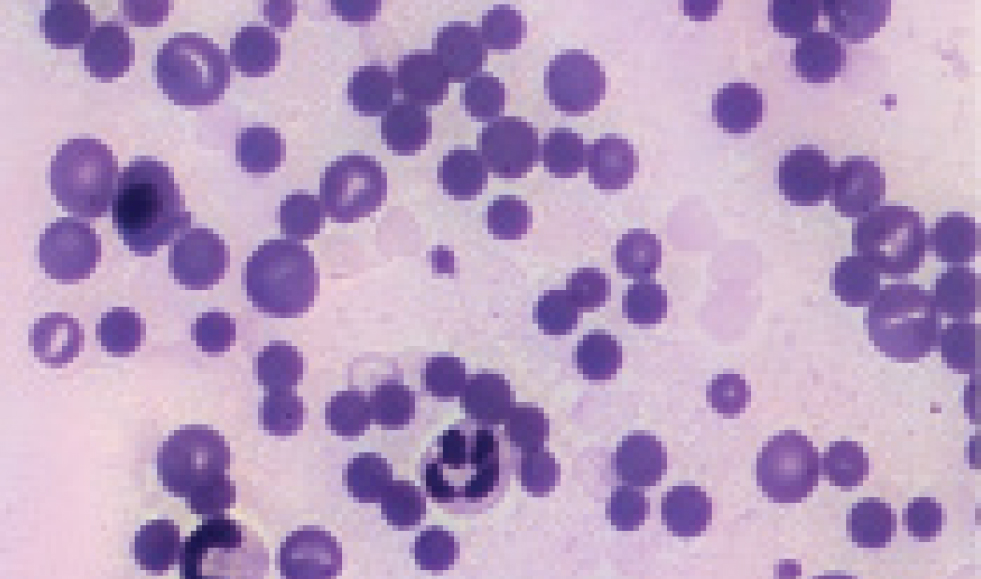
Once anaemia has been detected in a patient, the next step is to evaluate findings associated with immune-mediated destruction. This assessment includes findings such as spherocytosis and conducting tests such as the saline agglutination test and the direct antiglobulin test (Coomb's test) (Figure 3). A definitive diagnosis of immune-mediated haemolytic anaemia is typically confirmed when at least two of these tests yield positive results, or when a positive saline agglutination test persists even after washing (Swann et al, 2019). Spherocytes are thought to be produced when partial phagocytosis of the red blood cell occurs as a result of immunoglobulin G coating the cells (Weiss and Wardrop, 2010). However, spherocytosis in cats is difficult to assess as feline red blood cells do not consistently display central pallor (Garden et al, 2019). The saline agglutination test is an easy patient-side test which is helpful in both dogs and cats. Saline agglutination testing performed by mixing 4 drops of saline with 1 drop of blood has a reported specificity of 100% for immune-mediated haemolytic anaemia in dogs (Paes et al, 2013). If the saline agglutination test is negative, then a direct Coomb's test is advised.

Haemolysis
Signs of haemolysis include hyperbilirubinaemia, haemoglobinaemia, haemoglobinuria and ghost cells. At least one of these should be present for a firm diagnosis (Swann et al, 2019). In dogs, spherocytes can provide evidence of haemolysis. In the absence of decreased functional hepatic mass, obstructive cholestasis or sepsis, hyperbilirubinaemia may indicate haemolysis. Haemoglobinaemia is another indication of haemolysis and can be detected by visual examination of plasma or measurement of cell‐free haemoglobin (Weiss and Wardrop, 2010). Haemoglobinaemia should only be interpreted as evidence of haemolysis after eliminating artifactual haemolysis. Common causes of in vitro haemolysis include, but are not limited to, traumatic venipuncture, freezing and storage. Assessment of haemoglobinuria should be performed using a fresh urine sample, and the likelihood of erythrocyte lysis in urine is anecdotally increased in alkaline, poorly concentrated or hyposthenuric urine samples. Ghost cells provide evidence of intravascular haemolysis if they are seen on a smear made immediately after blood collection (Figure 4) (Swann et al, 2019).
Primary vs secondary immune-mediated haemolytic anaemia
Primary immune-mediated haemolytic anaemia occurs when there is no inciting cause for the haemolysis, and treatment is aimed at suppressing the immune system. However, it is essential that secondary causes, such as infections, drugs and neoplasia, are ruled out. The most common infections resulting in secondary immune-mediated haemolytic anaemia are Babesia in dogs and haemotrophic Mycoplasma species in cats (Swann et al, 2019). It is essential that treatment is directed at specific infectious causes in order to successfully treat the secondary immune-mediated haemolytic anaemia. A thorough history is important to rule out possible drugs which can cause immune-mediated haemolytic anaemia, and thorough imaging (thoracic and abdominal computed tomography or thoracic radiographs and abdominal ultrasound) are required to rule out secondary neoplastic causes (Swann et al, 2019).
Treatment
In severely affected cases, a blood transfusion can be lifesaving. There is no specific packed cell volume at which a transfusion should be considered, and the decision should be based on the patient's clinical presentation. Patients that are haemodynamically unstable (tachycardia, poor pulses) should be considered candidates for transfusion. In the UK, the Pet Blood Bank can supply packed red blood cells (Garden et al, 2019), and whole blood from in-clinic donor dogs can also be considered. There is no UK blood bank for cats, and in-clinic donor cats are often relied on. As such, whole blood is most commonly transfused to cats.
Steroids provide the cornerstone to treatment of primary immune-mediated haemolytic anaemia (Swann et al, 2019). In dogs, prednisolone is generally given at 2 mg/kg orally either as a single dose or divided into two daily doses. For dogs >25 kg, it is advised to give 50 mg/m2 orally of prednisolone to avoid over-treating. In cats, prednisolone can be given at 2–4 mg/kg orally as a single dose or divided into 2 daily doses. In patients that are anorexic and severely affected, intravenous dexamethasone is preferred, given at a dose of 0.2–0.4 mg/kg/day (Swann et al, 2019).
Second-line immunosuppressive agents are often used in conjunction with corticosteroids to enhance therapeutic efficacy while minimising steroid-related side effects, such as polyuria, polydipsia, muscle wasting and increased risk of infections. By targeting different immune pathways, these drugs help reduce the overall corticosteroid dose required to achieve remission. The preferred second immunosuppressant in veterinary medicine has not been established (Swann et al, 2019).
Common second-line options include cyclosporin, which inhibits T-cell activation, and mycophenolate mofetil, which suppresses purine synthesis in lymphocytes. In dogs, azathioprine is an additional option, working via DNA synthesis inhibition, but it should never be used in cats because they are unable to metabolise the drug safely. The ACVIM consensus statement recommends introducing a second agent when initial response to corticosteroids is inadequate, when side effects are intolerable or for long-term maintenance therapy to allow gradual corticosteroid tapering while preventing relapse (Swann et al, 2019).
Human intravenous immunoglobulin is created by purifying of large volumes of donor plasma. One study using high-dose immunoglobulin M-enriched human intravenous immunoglobulin found that it is well tolerated (Bestwick et al, 2022a); however, small scale studies have been conflicting, with some finding no benefit in dogs with immune-mediated haemolytic anaemia and others finding that it may be useful for short-term stabilisation of some dogs with immune-mediated haemolytic anaemia because of significant increases in haematocrit, but did not appear to improve long-term survival (Bestwick et al, 2022a). Despite the lack of studies and scientific evidence, it is still proposed as a salvage option in dogs not responding to treatment with two immunosuppressive drugs (Swann et al, 2019).
If a poor response is seen to dual immunosuppressive therapy with or without intravenous immunoglobulin transfusion, then the options include adding in a third immunosuppressive medication or considering splenectomy. Studies using three immunosuppressive medications concurrently in immune-mediated haemolytic anaemia are lacking, but it has been shown in renal transplant cases that there is a high risk of side effects and opportunistic infections when three immunosuppressive drugs are used (Gregory et al, 2006), so patients must be monitored closely if triple therapy is used. If three medications are concurrently used, the ACVIM consensus statement recommends using drugs that target different immune pathways which would preclude concurrent administration of azathioprine and mycophenolate mofetil (Swann et al, 2019).
Splenectomy for patients with immune-mediated haemolytic anaemia has been poorly studied. Previous research has found it to be beneficial in dogs with immune-mediated thrombocytopenia (Horgan et al, 2009), but the benefit of splenectomy in cases of immune-mediated haemolytic anaemia could not be determined in one study (Bestwick et al, 2022b). However, splenectomy was beneficial in patients with immune-mediated haemolytic anaemia in another study (Horgan et al, 2009).
Thromboembolic disease
Thromboembolism is a significant risk, with the potential to cause death in dogs with immune-mediated haemolytic anaemia (Carr et al, 2002). Carr et al (2002) found that thromboembolism was present in 20 of 25 dogs given postmortem examinations. Cats are not known to be at increased risk of thromboembolism, so thromboprophylaxis is only required for canine patients (Swann et al, 2019).
The terms ‘thromboprophylaxis’ and ‘antithrombotics’ encompass antiplatelet medications, which are intended to impede platelet function (primary haemostasis), and anticoagulant medications, which are designed to hinder the activity of clotting factors (secondary haemostasis). Considering the underlying mechanisms of venous thromboembolism commonly seen in dogs with immune-mediated haemolytic anaemia, it is proposed that a treatment strategy incorporating anticoagulants may be the favoured approach for thromboprophylaxis, especially within the initial two weeks following diagnosis (Helmond et al, 2010). In cases where the use of anticoagulants, along with the necessary monitoring, is not accessible or practical, recommendations lean towards administering antiplatelet drugs as a preferable alternative to not using any form of antithrombotic medication. There is insufficient evidence to make strong recommendations on the choice of anticoagulant in immune-mediated haemolytic anaemia. The strongest evidence supports the use of individually dose‐adjusted unfractionated heparin (Helmond et al, 2010). Other anticoagulants, including enoxaparin and rivaroxaban, appear to be safe and may be efficacious (Swann et al, 2019). It is important to emphasise that unfractionated heparin should not be administered without individualised dose adjustment. In situations where this is not possible or practical, the use of injectable low-molecular-weight heparins or direct oral Xa inhibitors like rivaroxaban is suggested (Swann et al, 2019).
It is recommended that thromboprophylaxis be provided for all dogs with immune-mediated haemolytic anaemia, except those with severe thrombocytopenia (platelet count <30 000/ul) (Swann et al, 2019). A higher risk for thrombosis was found with increased alkaline phosphatase (P=0.042), increased bilirubin (P=0.047) and decreased albumin (P=0.012) (Carr et al, 2002). Clopidogrel, when used alone or in combination with ultra-low dose aspirin, was effective, safe and had similar short-term survival compared with ultra-low dose aspirin alone in a small group of dogs with primary immune-mediated haemolytic anaemia able to tolerate oral medications and treated with standard immunosuppressive treatment (Mellett et al, 2011). Further guidance on the use of antithrombotic medications in dogs is available in the Consensus on the Rational Use of Antithrombotics in Veterinary Critical Care guidelines (Goggs et al, 2019).
Tapering medication
It is expected that therapy will last for around 3–6 months. In the majority of cases, immunosuppressive therapy can be tapered and stopped; however, in some cases, continuous therapy (usually at a lower dose) is required to keep patients in remission (Swann et al, 2019). Prednisolone is the cornerstone of therapy, and the dose should not be reduced within the first 2 weeks. If the haematocrit is stable and >30% for 2 weeks after commencing therapy with improvement in the majority of measures of disease activity (including spherocytosis, agglutination, serum bilirubin concentration and reticulocyte count), it is recommended to decrease the dosage of prednisone or prednisolone by 25% (Swann et al, 2019). The prednisolone dose can then be reduced by 25% every 2–3 weeks thereafter if patients are stable. It is essential that haematology (ideally including blood smear assessment) is performed before each dose reduction.
Prognosis
In cats, immune-mediated haemolytic anaemia has been associated with a mortality rate of 23%. Relapses were reported in 31% cases (Kohn et al, 2006). In dogs, 50–88% will survive to discharge. Increased mortality has been found in dogs with hyperbilibrubinaemia, thrombocytopenia, hypoalbuminaemia and increased band neutrophil count. Patients with immune-mediated haemolytic anaemia can do well, but often require intensive treatment and very close monitoring.
Conclusions
Immune-mediated haemolytic anaemia is a complex, life-threatening disorder that requires early recognition and intervention to improve patient outcomes. Advances in diagnostic testing, including improved identification of regenerative responses and immune-mediated destruction, allow for earlier and more accurate diagnosis. Corticosteroids remain the mainstay of treatment, but second-line immunosuppressants and thromboprophylaxis play a critical role in improving survival rates. While blood transfusions can be necessary for stabilisation, they should be viewed as supportive therapy rather than a cure.
The most recent ACVIM classification system, which differentiates spontaneous (non-associative) immune-mediated haemolytic anaemia from disease-associated (associative) immune-mediated haemolytic anaemia, provides better clarity and guidance for clinical decision-making. Prognosis varies, with early diagnosis and appropriate management being key predictors of success. Future research should focus on optimising treatment protocols, reducing steroid-related side effects and identifying better immunosuppressive strategies to improve long-term survival and quality of life in affected animals.


